An article by Niamh McGuinness, Senior Analyst, CTT, Privacy Analytics
Just over a year ago, Health Canada stipulated that all clinical trials documents needed to make their way onto its designated portal. And, like those entering a portal in popular fiction, everyone in the industry wondered what the effect would be, what the future would look like.
Today, a little over one year in, it’s worth looking at precisely where this portal has taken us. Where are we now? Where will we go from here?
Need a refresher? Read our Early Impact of Health Canada’s New Guidelines
Health Canada PRCI dossiers: a year in review
At the time of writing this article, 41 dossiers had been published on the Health Canada Portal. Of these 41, 36 described their anonymization methodologies. Let’s look at these 36:
What dossiers have been published?
1. 50:50
To date, there is an even split between on-request releases of past drug submissions and new drug submissions (Figure 1).
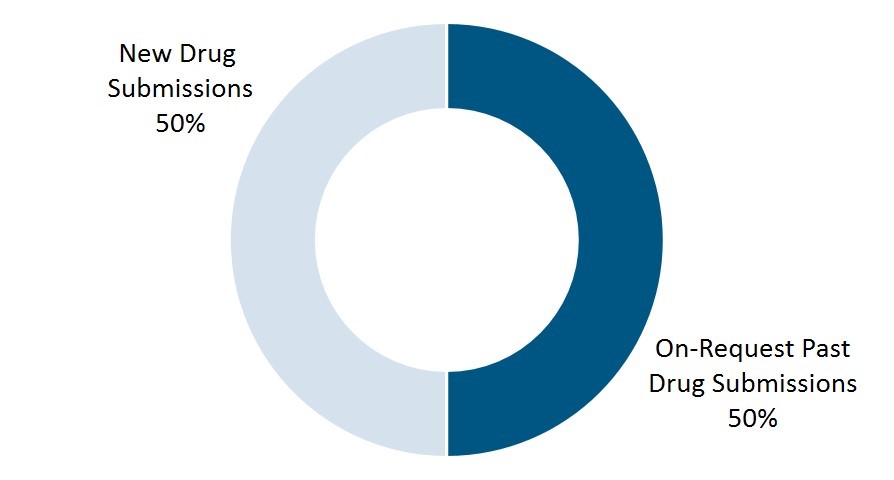
Figure 1. Proportion of new versus on-request drug submissions published to date
2. High interest in high cholesterol
The most common indication represented was hypercholesterolemia, or high cholesterol; 19% of all 36 submissions published were concerned with the drugs, called statins, that treat this condition. Of these releases, seven were on-request, accounting for 39% of past submissions. The degree of public interest in these drugs is unsurprising given that as many as one in ten Canadian adults are taking statins3.
3. Decades-old documents and submission feasibility
These formats may prevent effective automation of redaction and will likely preclude data transformation entirely.
This reality raises an important concern about the feasibility of timelines for on-request submissions. While sponsors can anonymize documents used in a new drug submission before the 60-day timeline – i.e., between Process Initiation and when the Submission of Annotated Documents begins – this is not the case with an on-request submission.
It’s becoming clear that 60 days for redaction of older dossiers or, indeed, large studies with numerous clinical reports, can be a challenging timeline.
What anonymization methods are being used?
1. An explicit preference for quantitative assessment
Both EMA and Health Canada have stated an explicit preference for a quantitative assessment of re-identification risk, with Health Canada elaborating further and stating that such an approach “has the advantage of being based on empirical measurement and consequently is more precise, less subjective, and typically retains more data utility.”
“… the advantage of being based on empirical measurement and consequently is more precise, less subjective, and typically retains more data utility.”
Out of the 125 dossiers on the EMA Portal containing identifiers, a qualitative risk assessment or fulfillment of the three criteria in the guidance—no possibility to a) single out, b) link records or c) infer information about individuals—was performed on 88% of dossiers. A quantitative risk assessment was performed on 12% of dossiers.
However, despite this stated preference, and the fact that both anonymization vendors and sponsors are maturing in their transparency capabilities, there has not been a dramatic increase in the use of risk-based anonymization to date on the Health Canada Portal.
2. Health Canada sees more quantitative approaches than the EMA
While Health Canada sees a stronger representation of statistical approaches to re-identification risk assessment than the EMA, these dossiers still account for only 14% of submissions—or 20% of dossiers if those reflecting a combination approach are included. (See Figure 2.)
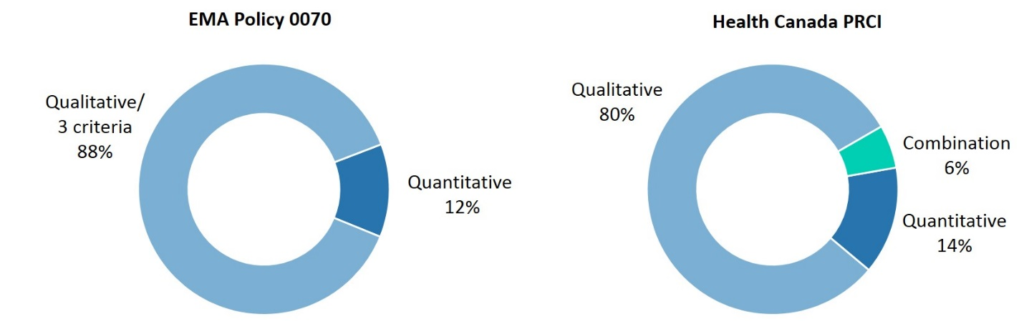
Figure 2: Methods used to assess patient re-identification risk under EMA Policy 0070 and Health Canada PRCI
3. The impact of redaction on data utility
Another key consideration in both policies is the mechanism by which the patient information is transformed, which also impacts the utility of the de-identified information.
In the majority of cases on both the EMA and Health Canada Portals, redaction is used as a means of masking patient information, accounting for 89% of dossiers published under Policy 0070 and 86% of those published in on the Health Canada Portal.
Therefore, a minority of dossiers have been subject to anonymization, or a combination of anonymization and redaction, as a means of protecting patient privacy.
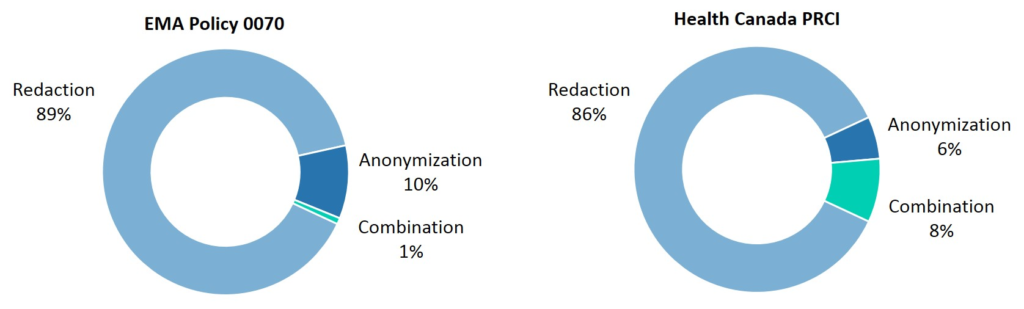
Figure 3: Methods used to protect patient privacy in the first year of EMA Policy 0070 and Health Canada PRCI
Defining redaction and anonymization
Redaction in this case means complete concealment of patient data with an opaque box, such that all of the inherent usefulness of the information is effectively removed.
While there may be no sensible alternative to redaction in the case of older dossiers under the Health Canada on-request provision, the concealment approach has been heavily relied-upon in the last two decades, with redaction occurring in 84% of documents published after 1999.
On the other hand, anonymization here refers to the replacement of the original text with resynthesized values selected to bring the re-identification of a given trial participant below a given threshold.
The aim of anonymization is to maintain as much data utility and referential integrity as possible, and uses a combination of methods. These may include, but are by no means limited to, pseudonymization of direct identifiers, generalization of medical histories and ages, and date offsetting according to the PhUSE standard4. A visual comparison is shared below.

What does this mean for transparency leaders?
1. Does a clear preference for anonymization presage its inevitable adoption?
Both the EMA and Health Canada assert that a quantitative approach to measuring re-identification risk, coupled with data transformation methods that preserve data utility, should be used wherever possible1,2. Not only are these methods consistent with regulatory preferences, they have also been demonstrated to withstand commissioned re-identification attacks 7.
While redaction is still acceptable to Health Canada at this time, even for machine-readable documents, it is possible that this will be phased out in the next stages of implementation of the initiative.
EMA Policy 0070 activities have been on hold since August 2018—with no indication of when they may continue.
2. Where will the Health Canada Portal lead from here
One of the takeaways from the first year of Health Canada’s PRCI is that public engagement with clinical trial transparency efforts is high, with half of all dossiers being published on an on-request basis.
As we move into Year Two of the initiative, and as more regulators globally move towards adopting similar measures, we may also see an increase in . It is logical that this will necessitate enhanced efforts to achieve the richest possible data while protecting patient privacy—a balance which is most effectively struck using quantitative risk-based anonymization.
Both portals—Health Canada’s and the EMA’s—will soon show us more of the future.
References
[1] European Medicines Agency policy on publication of clinical data for medicinal products for human use, 21 March 2019, EMA/144064/2019.
[2] Public Release of Clinical Information: guidance document, v1.0, March 12 2019.
[3] Hennessy, D. A., Tanuseptro, P., Tuna, M., Bennett, Co., Perez, R., Shields, M., Ko, D. T., Tu, J., Manuel, D.G. (2016) Population Health Impact of statin treatment in Canada, Health Reports, Statistics Canada, Catalogue no. 82-003-X
[4] PhUSE De-Identification Working Group. De-identification standards for CDISC SDTM 3.2. Technical report, PhUSE, 2015.
[5] Ferran, J-M., Nevitt, S. (2019) European Medicines Agency Policy 0070: an exploratory review of data utility in clinical study reports for academic research, BMC Medical Research Methodology, 19:204
[6] Branson, J. et al. (2020), Evaluating the re-identification risk of a clinical study report anonymized under EMA Policy 0070 and Health Canada Regulations, Trials, 21, 200.